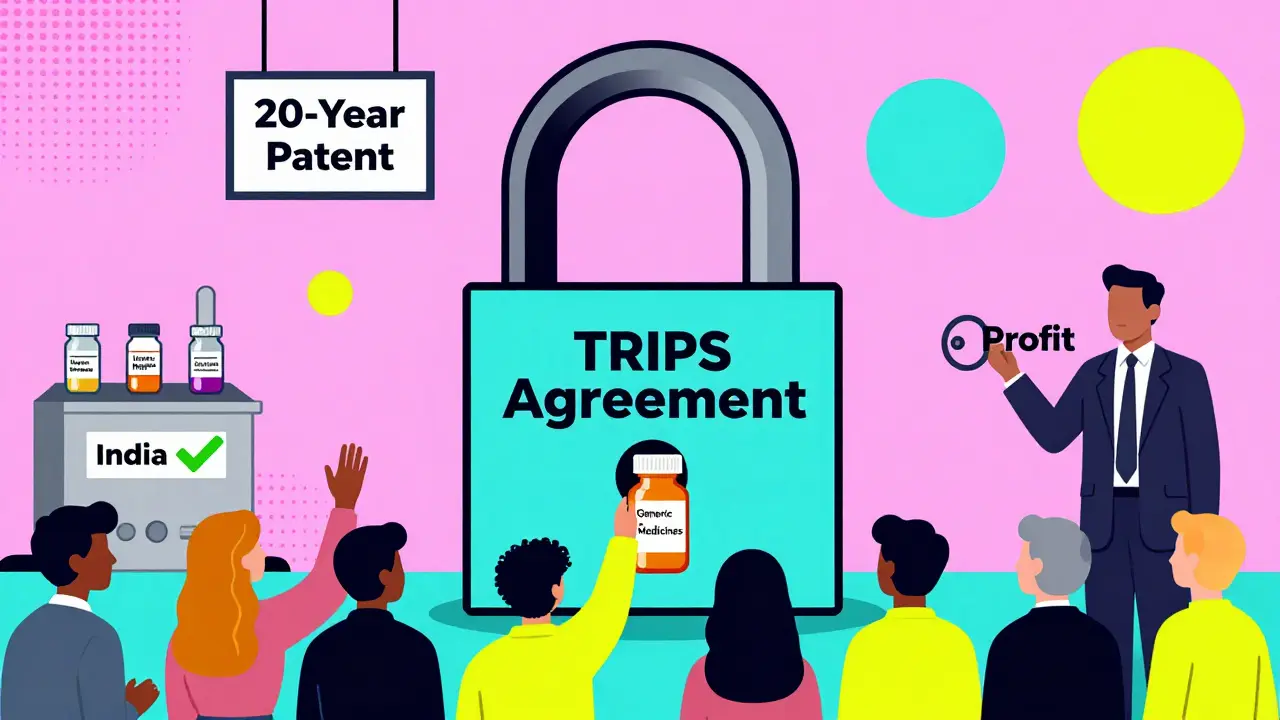
When you need a life-saving drug, the price shouldn’t depend on which country you live in. But for millions of people in low- and middle-income countries, the cost of medicine is locked in by a global rulebook written decades ago: the TRIPS Agreement is a World Trade Organization treaty that sets minimum standards for intellectual property protection, including 20-year patents on pharmaceuticals. Also known as the Trade-Related Aspects of Intellectual Property Rights Agreement, it came into force in 1995 and reshaped how medicines are made, sold, and accessed worldwide.
What TRIPS Changed for Generic Drugs
Before TRIPS, many developing countries didn’t patent drugs at all. India, for example, allowed companies to make generic versions using different manufacturing methods - even if the original drug was patented elsewhere. This kept prices low. A 1995 WHO survey found only 23 of 102 developing countries granted product patents for medicines. By 2010, that number jumped to 147. Why? Because TRIPS forced every WTO member - from Brazil to Bangladesh - to adopt strict patent rules.
The agreement requires all members to grant patents for at least 20 years from the filing date. That means even if a drug was invented in the U.S. in 2005, no one else can legally make it until 2025 - no matter how many people need it. And it’s not just patents. TRIPS also introduced data exclusivity, which blocks generic manufacturers from using the original company’s clinical trial data to get approval. Even after the patent expires, this can delay generics by 5 to 10 years.
The result? Prices skyrocketed. A 2001 study in the Journal of the American Medical Association found that patented drugs in developing countries cost, on average, over 200% more after TRIPS was enforced. In South Africa, when the government tried to import cheaper HIV drugs in 1998, 40 pharmaceutical companies sued. It took global protests and media pressure just to get them to drop the case.
Compulsory Licensing: The Legal Loophole
TRIPS isn’t completely rigid. Article 31 lets governments issue compulsory licenses - legal permissions for someone else to produce a patented drug without the company’s consent. This is meant for emergencies: pandemics, HIV outbreaks, or when a medicine is unaffordable.
Thailand used it in 2006 to make generic versions of HIV and heart drugs. Brazil did the same for antiretrovirals. Both faced intense pressure from the U.S. and drug companies. But they held. The problem? TRIPS says these licenses must be used mostly for the domestic market. So if a country doesn’t have a drug factory, it can’t import generics made in another country under a similar license.
That changed in 2005 with the so-called “Paragraph 6 Solution.” It allowed countries without manufacturing capacity to import generics made under compulsory license. Sounds fair, right? In practice, it was a nightmare. The paperwork was complex. Few countries knew how to use it. By 2016, only one shipment of malaria medicine had ever been sent under this rule. Canada and Rwanda were the only two countries to fully use it - and even then, only after years of legal maneuvering.
TRIPS Plus: The Hidden Rules
TRIPS is just the baseline. Wealthier nations have been slipping in stricter rules through trade deals - what experts call “TRIPS Plus.” The U.S., EU, and Switzerland now routinely demand extra protections in bilateral agreements. These include:
- Extending patent terms beyond 20 years
- Imposing 8-10 years of data exclusivity (TRIPS only suggests 5)
- Banning compulsory licensing for certain diseases
- Forcing “patent linkage” - where drug regulators must check patent status before approving generics
A 2020 U.S. International Trade Commission report found that 85% of U.S. free trade agreements included TRIPS Plus provisions. The EU-Vietnam deal, signed in 2020, gave 8 years of data exclusivity - two years longer than TRIPS allows. These aren’t just technicalities. They’re barriers that delay generics by years, keeping prices high.

Who Wins? Who Loses?
The pharmaceutical industry argues that strong patents drive innovation. They point out that 73% of new medicines approved since 2000 came from companies in countries with strict IP laws. But here’s the catch: most of those drugs aren’t for the diseases killing millions in poor countries. Between 1975 and 1997, only 13 of 1,223 new drugs were developed for tropical diseases like sleeping sickness or leishmaniasis.
Meanwhile, the people who need these drugs most are priced out. The Global Fund reported that in 2000, treating HIV cost $10,000 per patient per year. By 2019, that dropped to $75 - thanks entirely to generic competition in countries that fought for access. But newer treatments, like those for hepatitis C or cancer, still cost $300-$600 per year because patents haven’t expired - or because TRIPS Plus rules block generics.
India’s transition to product patents in 2005 is a case study. Before, it was the “pharmacy of the developing world,” making 80% of the world’s generic HIV drugs. After 2005, prices for patented cancer drugs jumped 300-500%, according to a Lancet Oncology study. The country still makes generics - but only for drugs where patents have expired or don’t exist.
The COVID-19 Waiver and What It Means
In October 2020, India and South Africa proposed a temporary waiver of TRIPS for vaccines, tests, and treatments related to COVID-19. Over 100 countries supported it. The EU, U.S., and Switzerland resisted. It took over a year of global pressure before the WTO agreed to a partial waiver in June 2022.
It wasn’t perfect. The waiver only covered vaccines - not treatments or diagnostics. It was limited to developing countries. And it didn’t force companies to share technology. Still, it was historic. It was the first time the WTO modified TRIPS to prioritize public health over patents.
But the fight isn’t over. The same countries that blocked the waiver now push TRIPS Plus in new trade deals. Meanwhile, the Medicines Patent Pool - a UN-backed initiative - has negotiated licenses for 16 HIV drugs, 6 hepatitis C treatments, and 4 TB medicines. As of 2022, those deals reached 17.4 million people. That’s progress. But it’s a patchwork solution, not a system change.

The Real Cost of Patents
When a drug is patented, the company has a monopoly. That means they set the price. In the U.S., a single dose of insulin can cost $250. In Nigeria, it’s $10 - because it’s generic. The difference isn’t production cost. It’s patent law.
Between 1995 and 2015, the WTO-UNDP TRIPS Trust Fund spent $28.7 million helping 102 countries implement the agreement. But almost no money went to help them use the flexibilities - like compulsory licensing - to protect public health. That’s like giving someone a lock and then refusing to give them the key.
And the result? A 2015 World Bank analysis found that countries implementing TRIPS without public health safeguards saw a 15-20% drop in generic medicine availability within five years. That’s not a side effect. It’s the design.
What’s Next?
There are two paths ahead. One leads to more TRIPS Plus deals, tighter patent controls, and continued delays in generic access. The other leads to reform: expanding compulsory licensing, eliminating data exclusivity, and letting countries import generics freely.
The Medicines Patent Pool shows what’s possible when collaboration replaces control. But it’s not enough. The system still prioritizes profit over people. Until the rules change, millions will keep dying because the medicine they need is locked behind a patent - not because it doesn’t exist, but because the law says it can’t be made.
Does TRIPS allow countries to make generic drugs?
Yes, but with major restrictions. TRIPS permits compulsory licensing - allowing governments to authorize generic production without the patent holder’s permission - but only under strict conditions. The license must be non-exclusive, limited to the domestic market, and issued after attempts to negotiate a voluntary license fail. Only in emergencies can these rules be bypassed, and even then, cross-border trade is heavily limited.
Why do generic drugs cost so much in poor countries?
They don’t - if they’re allowed to be made. The high cost comes from patent laws that block generic production. Even after patents expire, data exclusivity and patent linkage rules prevent regulators from approving generics. In many countries, local manufacturers lack the legal or technical capacity to challenge these barriers. The result? Patented drugs remain expensive, while cheaper generics are delayed or banned.
What’s the difference between a patent and data exclusivity?
A patent gives the original company exclusive rights to make and sell the drug for 20 years. Data exclusivity is different: it blocks generic makers from using the original company’s clinical trial data to prove their version is safe and effective. Even if the patent expires, data exclusivity can delay generic entry for 5-10 more years. It’s a legal loophole that extends monopolies without a patent.
Can a country import generics made under compulsory license from another country?
Only under very limited conditions. The 2005 “Paragraph 6 Solution” allows it, but the process is complex. The importing country must prove it lacks manufacturing capacity, the exporting country must issue its own compulsory license, and both must follow strict reporting rules. Only one shipment of medicine - malaria drugs from Canada to Rwanda - has ever been sent this way. Most countries can’t or won’t navigate the bureaucracy.
Is TRIPS the reason new medicines aren’t developed for poor countries?
Not directly - but it reinforces the problem. The pharmaceutical industry focuses on profitable markets. Between 1975 and 1997, only 13 of over 1,200 new drugs were for tropical diseases. TRIPS doesn’t cause this, but it locks in a system where patents are only valuable if the market can pay. Poor countries can’t afford to fund R&D, so companies don’t invest. TRIPS makes it harder to fix that gap by blocking generic production.
What happened after the COVID-19 TRIPS waiver?
The June 2022 waiver allowed developing countries to produce and export COVID-19 vaccines without patent permission. But it was narrow: it only covered vaccines, not treatments or diagnostics. It didn’t require technology sharing. And it was temporary. While it set a precedent, it didn’t change the core rules of TRIPS. The same countries that blocked it are now pushing stronger patent rules in new trade deals.

14 Comments
Erica Santos
March 8, 2026 AT 05:05Oh wow, a 20-year monopoly on life-saving drugs? How quaint. I bet Pfizer’s boardroom has a golden toilet made from insulin vials. Let me guess - the real ‘public health crisis’ is that CEOs can’t afford to buy yachts anymore? 🤡
TRIPS isn’t a treaty - it’s a corporate welfare program disguised as international law. And don’t even get me started on ‘data exclusivity.’ That’s just patent law with extra steps to make sure people die slower.
They call it ‘innovation.’ I call it extortion with a PhD.
Meanwhile, in India, a man makes a generic version of a cancer drug for $2 a dose - and suddenly he’s a ‘pirate.’ But when Big Pharma hikes insulin to $300 in the U.S.? That’s ‘market dynamics.’
It’s not about patents. It’s about power. And the WTO? More like the World Trade Oligarchy.
Every time someone says ‘we need to protect innovation,’ I hear: ‘we need to protect profits while people gasp for breath.’
And let’s not pretend the COVID waiver was a victory. It was a PR stunt. One vaccine. No treatments. No diagnostics. No tech transfer. Just enough to make the Global North look like they gave a damn.
Meanwhile, the same countries pushing TRIPS Plus in Africa are now crying about ‘supply chain security’ - while blocking the very tools that could save 10 million lives.
It’s not capitalism. It’s cannibalism with a PowerPoint.
And you know what’s worse? The silence. The way people nod along like this is normal. Like it’s inevitable. Like it’s not a choice.
It’s a choice. And we’re choosing death.
George Vou
March 8, 2026 AT 07:06lol so u know what i think? its all a big lobbie by the fda and big pharma to keep prices high so they can buy more private jets
also i heard the un is run by aliens and they made tris so we all get mind controlled by vaccines
my cousin works at a pharmacy and he says the real reason generics are banned is because the gov wants us to be dependent on pills
and dont get me started on the 5g towers they put in hospitals to make people sicker so they buy more drugs
its all connected man
watch the documentary 'the medicine plot' on youtube - its 100% real
also i think the pope is in on it
Scott Easterling
March 9, 2026 AT 10:37Oh, here we go again. Another ‘TRIPS is evil’ post. Let me guess - you also think vaccines cause autism and climate change is a hoax? 🙄
Patents aren’t the problem. The problem is that developing countries can’t build infrastructure. They can’t train pharmacists. They can’t refrigerate insulin. Stop blaming patents for your own failures.
And ‘compulsory licensing’? Please. That’s just legal theft with a UN stamp. If you want drugs, build a factory. Don’t ask the world to hand you someone else’s R&D.
Also - ‘TRIPS Plus’? That’s just free trade. You think Europe and the U.S. should give away their intellectual property like it’s a charity? No. We work for it. You didn’t.
And don’t even mention India. They stole 80% of the world’s HIV drugs before 2005. That’s not ‘pharmacy of the world.’ That’s piracy.
Stop romanticizing theft. Innovation doesn’t happen in a vacuum. Someone paid for that research. Pay up.
And if you’re so outraged - why don’t you donate to the Medicines Patent Pool? Oh right. You’d rather just scream on Reddit.
Mantooth Lehto
March 10, 2026 AT 01:02I just cried reading this. 💔
I lost my brother to hepatitis C in 2017. The drug cost $94,000. I sold my car. I maxed out my credit cards. I begged my employer. Nothing.
Meanwhile, in Egypt? $200. Same drug. Same chemistry. Just… legal.
It’s not about money. It’s about dignity. And the system took that from him.
I’m not mad. I’m heartbroken.
And now? They’re pushing TRIPS Plus again. In Africa. In Latin America.
How many more brothers? How many more sisters?
Why are we still letting this happen?
Melba Miller
March 10, 2026 AT 19:18Let’s be clear: this isn’t about access. It’s about control. And the U.S. government? It’s complicit.
Every time they sign a trade deal with Vietnam or Colombia, they slip in TRIPS Plus like a knife in the back. Data exclusivity? Patent linkage? You think that’s for ‘fairness’? No. It’s to keep generic manufacturers in the Global South from ever catching up.
India’s generic industry? Built on decades of innovation. Not theft. Just different rules.
Now? They’re being forced into the same cage. And we’re celebrating it as ‘global harmonization.’
Harmonization? More like colonization with a spreadsheet.
And don’t tell me about ‘incentives.’ The U.S. spends $50 billion a year on drug R&D - and 80% of it comes from taxpayer-funded NIH grants.
So we’re paying twice. Once for the science. Once for the monopoly.
It’s not capitalism. It’s theft. And we’re the accomplices.
Katy Shamitz
March 12, 2026 AT 17:27Oh my goodness, this post made me so emotional 😭
I just want to say - I believe in compassion. I believe in healing. And I believe no one should have to choose between rent and insulin.
My cousin in Guatemala had to wait 18 months for her diabetes meds because the local pharmacy couldn’t afford the branded version.
That’s not a policy failure. That’s a moral failure.
We need to stop pretending this is about economics. It’s about love. About humanity.
Let’s support the Medicines Patent Pool. Let’s write our reps. Let’s demand change.
Because if we don’t, who will?
I’m sending you so much light and warmth 💛
Nicholas Gama
March 13, 2026 AT 10:38Patents fund innovation. No patents? No drugs. Simple.
TRIPS is the minimum. If you want generics, build your own labs.
Stop blaming the system. Build the system.
Mary Beth Brook
March 14, 2026 AT 16:48TRIPS Plus is a necessary evolution of IP governance in a globalized pharma landscape. The absence of harmonized data exclusivity regimes creates regulatory arbitrage, undermines R&D ROI, and incentivizes jurisdictional shopping - which distorts market efficiency and compromises clinical safety standards.
Compulsory licensing without technology transfer is symbolic. It doesn’t build capacity. It creates dependency.
The real solution? Public-private innovation partnerships. Not legal loopholes.
Neeti Rustagi
March 15, 2026 AT 11:04As someone from India, I have witnessed firsthand the power of generic manufacturing. Before 2005, our country produced 40% of the world’s HIV antiretrovirals. Not because we stole - but because we built an ecosystem of skilled chemists, engineers, and ethical manufacturers.
TRIPS did not ‘force’ us to change - it forced us to adapt. And we did - with dignity.
But the real injustice? The West never helped us build the capacity to innovate - only to copy. Now, they call us ‘cheaters’ for doing what they once did.
Let us not confuse moral courage with moral hypocrisy.
True justice is not in waiving patents - but in funding R&D for diseases that affect the poor.
Dan Mayer
March 15, 2026 AT 15:44wait so u mean the u.s. is like… not perfect? wow. mind blown. 😵
patents are what make medicine possible. if u take them away, no one will invent anything. duh.
also i think the WHO is run by china and they want us all to take ivermectin
and tris? more like tris-uh
also i read this one article once
Janelle Pearl
March 17, 2026 AT 06:03I just want to say - thank you for writing this. I’ve been working in global health for 15 years, and this is the most accurate summary I’ve seen.
What breaks my heart? The silence. The way people look away. The way we normalize suffering because it’s ‘too complicated.’
But it’s not complicated. It’s cruel.
I’ve held the hands of mothers in rural Kenya who couldn’t afford their child’s asthma inhaler. I’ve sat in clinics where the only drug on the shelf was expired.
Change is possible. We’ve done it before - with HIV. With hepatitis C.
It takes political will. Not just pity.
I’m not angry. I’m determined.
And I’m not giving up.
Ray Foret Jr.
March 17, 2026 AT 12:09man i just read this and i feel so inspired 😊
we can fix this. we really can.
im gonna donate to the medicines patent pool today
and im gonna write my senator
and im gonna tell my friends
change starts with us
love u all 💙
Samantha Fierro
March 18, 2026 AT 05:31This is a meticulously researched and deeply necessary analysis. The structural inequities embedded in TRIPS and its derivatives are not incidental - they are systemic.
The failure of the WTO to meaningfully implement Paragraph 6, the absence of binding technology transfer mechanisms, and the deliberate marginalization of public health flexibilities reveal a governance architecture designed to serve capital, not conscience.
Reform requires more than moral appeals. It requires legal innovation, multilateral enforcement, and the dismantling of the myth that intellectual property is a neutral category.
Thank you for this contribution.
Robert Bliss
March 18, 2026 AT 19:01Just wanted to say - I’m not a doctor or a lawyer. But I get it.
If someone I love needed a drug, and I could get it for $20 instead of $2000… I’d take it.
And I wouldn’t feel guilty.
Why should anyone else?
Thanks for sharing this. It matters.
💙