ClearSkyPharmacy.Biz: Your Trusted Source for Pharmaceuticals - Page 2
FDA Inspection of Generic Manufacturing Facilities: What to Expect in 2026
Learn what to expect during an FDA inspection of generic drug facilities. Covers the 6-system approach, Form 483 handling, and the new PreCheck program for 2026 compliance.
Antipsychotics and Stroke Risk in Seniors with Dementia: What Doctors Won’t Tell You
Antipsychotics for dementia-related behavior increase stroke risk by up to 80% - even after just a few days. Learn why these drugs are still prescribed despite deadly risks, and what safer alternatives actually work.
Compulsory Licensing: How Governments Override Patents to Save Lives
Compulsory licensing lets governments override patents to make life-saving drugs affordable. Used in India, Brazil, and Thailand, it cut drug prices by up to 90%. A legal tool under international law, it balances innovation with public health needs.
Mindfulness for Anxiety: Proven Techniques and Clinical Evidence
Mindfulness for anxiety is backed by clinical evidence showing it can be as effective as medication without side effects. Learn how MBSR and MBCT work, what the science says, and how to start today.
How to Report Adverse Events to the FDA for Medications
Learn how to report medication side effects to the FDA through MedWatch. This guide covers who should report, what info to include, how the system works, and how your report helps protect public health.
Therapeutic Equivalence: What It Really Means for Patient Safety
Therapeutic equivalence ensures generic drugs perform just like brand-name ones, with the same safety and effectiveness. It's not just about cost - it's a science-backed system that protects patient health.
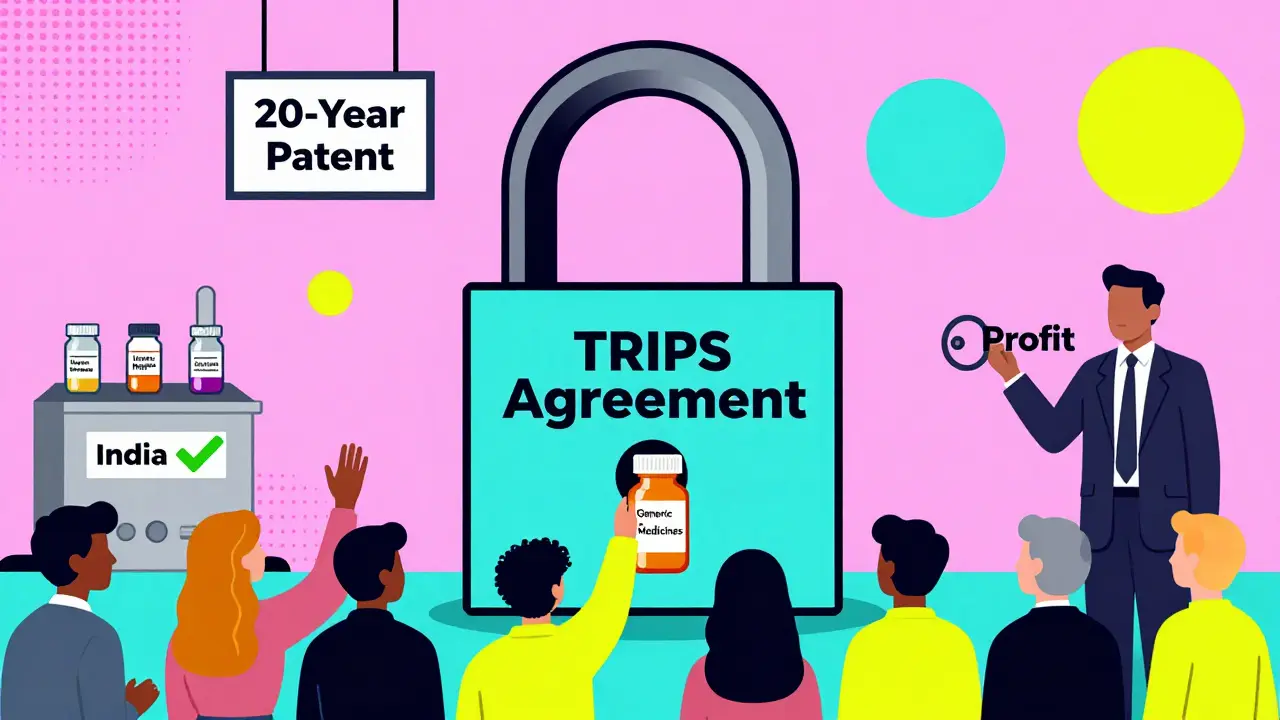
How TRIPS Agreement Shapes Global Access to Generic Medicines
The TRIPS Agreement governs global patent rules for medicines, blocking generic production in many countries. It has raised drug prices, delayed access, and created legal barriers - even as lifesaving generics exist.
Verbal Prescriptions: Best Practices for Clarity and Safety
Verbal prescriptions are still used in emergencies and surgeries, but they carry high risks of error. Learn the proven safety practices - like read-back and phonetic spelling - that prevent deadly mistakes.
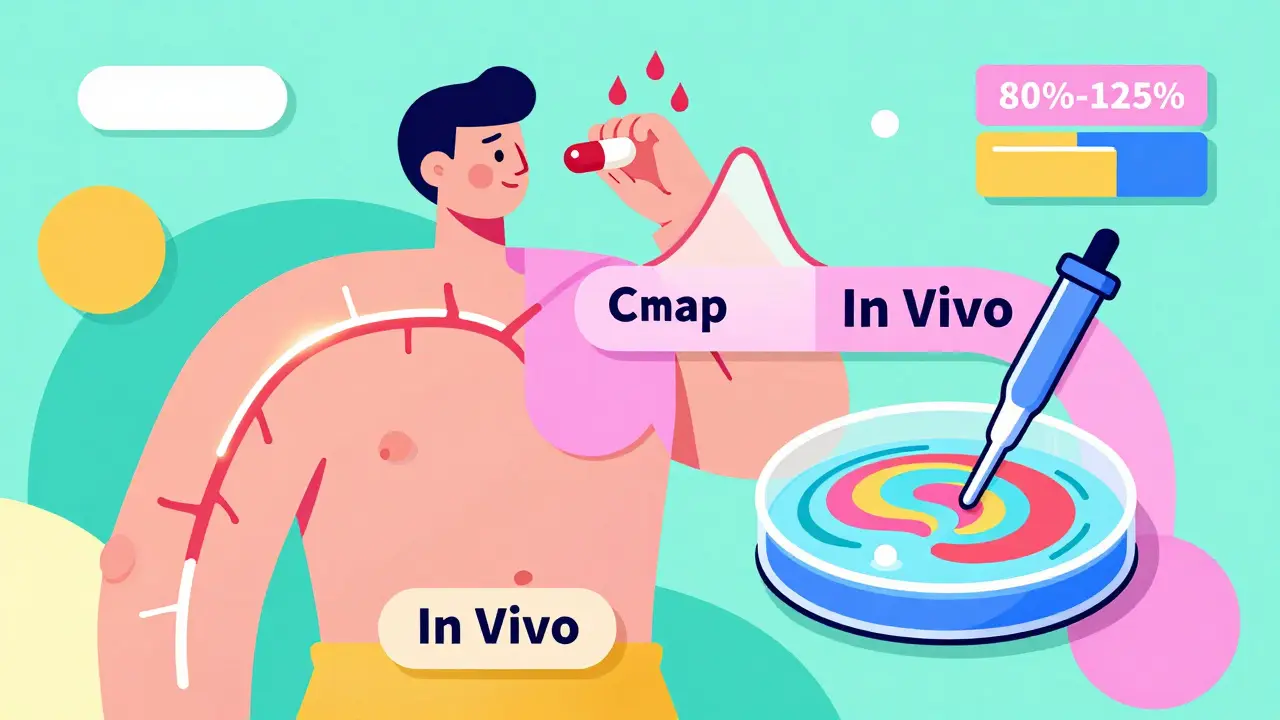
In Vivo vs In Vitro Bioequivalence Testing: When Each Is Used
In vivo and in vitro bioequivalence testing determine if generic drugs work like brand-name versions. In vivo uses human trials; in vitro uses lab tests. Learn when each is required and why regulators are shifting toward lab-based methods.
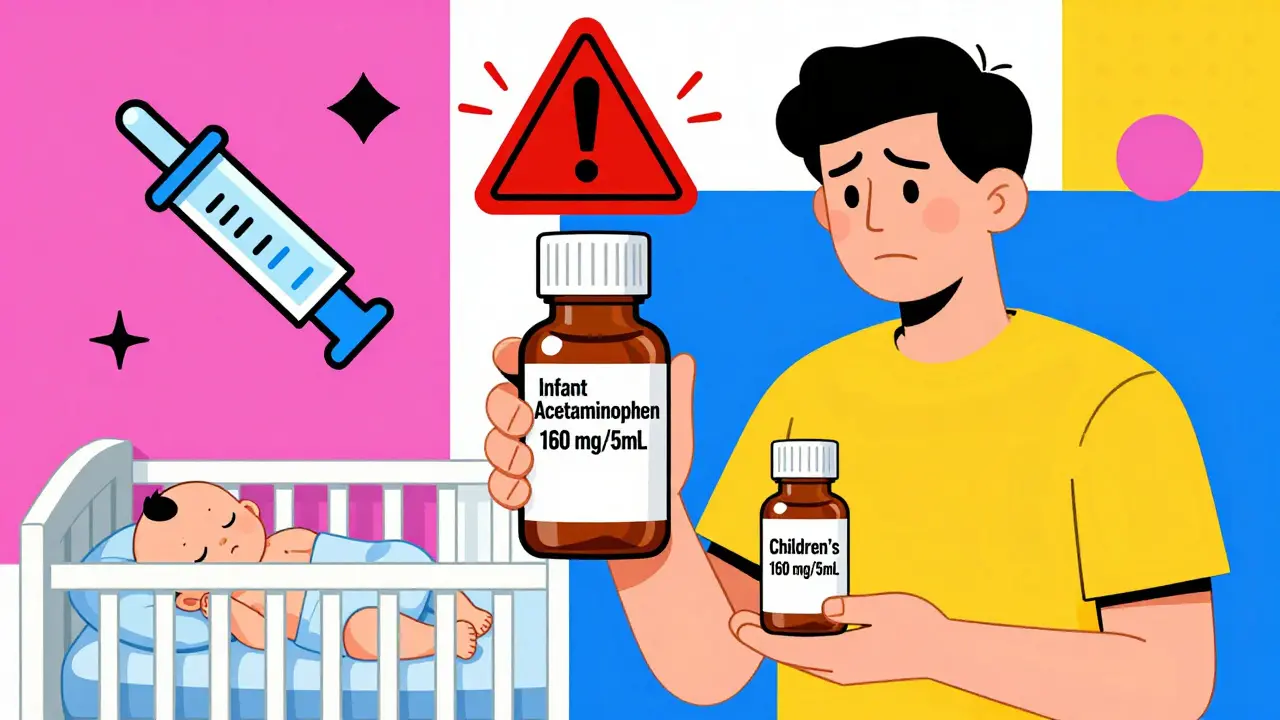
Infant Medication Safety: Drops, Concentrations, and Dosage
Infant medication errors cause thousands of ER visits each year. Learn how to safely measure drops, understand concentrations, and avoid deadly mistakes with oral syringes and accurate dosing.
© 2026. All rights reserved.